Calculate the Value of the Rate Constant K
Therefore the units of k assuming that concentration is represented in molL-1 or M and time is represented in seconds can be calculated via the following equation. Show your calculations including units.

Determine The Rate Constant K For A Reaction Youtube
Round your value for the rate constant to 3 significant digits.
. Calculate the average experimental value for k. Calculate the frequency factor A. At 20C 293 K the value of the fraction is.
Question Using the data in the table calculate the rate constant of this reaction Using the data in the table calculate the rate constant of this reaction. Rate constant from the Arrhenius equation. The following solution is suggested to handle the subject Using the data in the table calculate the rate constant of this reaction.
From the Arrhenius equation it is apparent that temperature is the main factor that affects the rate of a chemical reaction. 8 How do you calculate rate constant. It must be worked out from the rate equation 2.
The ratio of the rate constants gives us a new constant the equilibrium constant K which is defined as follows. T K k s-1 658 214105. K A e E a R T.
Calculating K from Initial Amounts and One Known Equilibrium Amount. 1 Exp 4 0043 OR 43 102 OR 0044 OR 44 102 They can score all 3 if they have used their incorrect value of k. M-1 Share Improve this answer.
Also be sure your answer has the correct. How to Find the Rate Law and Rate Constant k - YouTube. That causes the rate of reaction to almost double.
Lets keep an eye on the content below. Kdfrack_fk_r labelEq5 Hence there is a fundamental relationship between chemical kinetics and chemical equilibrium. Write the equilibrium expression for the reaction.
By raising the temperature just a little bit to 303 K this increases. Exp 2 rate k 10125 104 Exp 3 Q 002k Exp 4 P 00913k 1. The activation energy of a reaction is 225 k Cal mol1 and the value of rate constant at 40C is 18 105 s1.
Here A is a constant for the frequency of particle collisions Ea is the activation energy of the reaction R is the universal gas constant and T is the absolute temperature. A and B are the molar concentration of reactants A and B. 11 What is collision factor.
The value of k refers to a specific temperature and it increases if we increase temperature. Remember that the solutions were diluted. If three wrong answers check their value of k in b.
Include the correct units. 1 Values of the rate constant for the decomposition of N2O5 at four different temperatures are as follows. It is constant at a fixed temperature.
K Ae -EaRT. The concentration of nitrogen dioxide starts at zero and increases until it stays. Rate constant of first order reaction is the constant of proportionality to initial concentration and the amount of the reactant reacted or product formed and is represented as K ln C 0 C 0-x t or Rate constant ln Initial concentration Initial concentration-Amount reacted in time t Reaction Time.
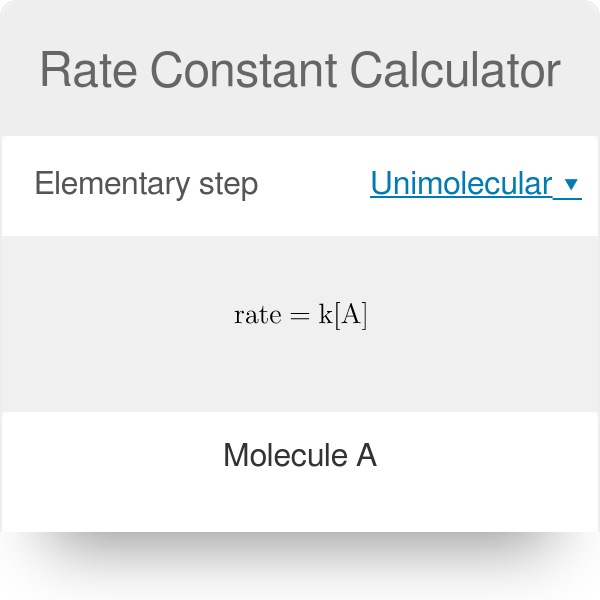
4 What is the rate constant at 760 K. Now we can continue onwards as follows. K RateA x B y.
Determine the molar concentrations or partial pressures of each species involved. 9 How do you find the rate constant for a reaction. Use this information to write a rate law for this reaction and calculate the value of the rate constant k.
R a t e A a B b. The value of the gas constant R is 831 J K-1 mol-1. Following are the ways to express rate constant.
6 How do you find rate constant from temperature. Write the complete rate law inserting the appropriate numerical values in place of the vari- ables k x and y. K_textobs k times textbleach Leftrightarrow k frack_textobstextbleach 00299mathrms-1.
The rate of a certain reaction increases by 23 times when the temperature is raised from 300 K to 310 K. The units of k depend on the overall order of reaction. The preexponential factor A and the temperature T must be given.
0 1 0 2 k J m o l 1. The calculations are done after the input of each value however perfoming effectively only those calculations that are possible with the input values present. If k is the rate constant at 300 K then the rate constant at 310 K will be equal to.
See the answer See the answer done loading. A graph with concentration on the y axis and time on the x axis. The elimination rate constant is the rate at which drug is cleared from the body assuming first-order elimination.
This is the value in the rule-of-thumb often used. 7 How do you find the rate constant from a table. We can graph the concentration of and over time for this process as you can see in the graph below.
The activation energy of the decomposition reaction is 102102 kJmol. A and b are the order of the reaction. You can see that the fraction of the molecules able to react has almost doubled by increasing the temperature by 10C.
Rearranging the rate equation the value of the rate constant k is given by. Where k is the rate constant. What is the value of A when T.
Rate constant k 2 1 0 3 m o l 1 L s 1 a n d E a 2. Calculate the value of the rate constant at 300K. K Ms-1M-n M 1-ns-1.
This tool may be used to calculate the temperature dependent values of a 1. Under a given set of conditions the composition of the equilibrium mixture is determined by the magnitudes of the rate. Trial AM BM Rate Ms 1.
Various abbreviations are used to represent the elimination rate constant including k e k el λ and λ zThe calculation of the elimination rate constant can be done using pharmacokinetic parameters or it can be done directly from a plot of concentration time. Initial concentration of the sample before diffusion or reaction. B Calculate the value of the rate constant k for the reaction.
Using Arrheneius equation we getlnk RTE a ln k 2 k 1 RE a T 2 1 T 1 1 on substitution we getln3510 4510 4 1987E a 3201 3001 E a 1047812 calmolRate constant at 37 C T310 Klnk 19873101047812 1701 k40910 8. Calculate the value of the true rate constant k from the experimental value for k Trial II. Order chemical rate constant following the equation k A TT R B exp-E A RT.
Substitute each concentration into the equilibrium expression and calculate the value of the equilibrium constant. Include the correct units. 5 What is the numerical value of the rate constant k.
10 How do you find the rate. Concentration of A squared r kA2 The rate constant k 1. The value of k is independent of concentration and time.

How To Determine The Units Of The Rate Constant K Chemical Kinetics Youtube

Comments
Post a Comment